 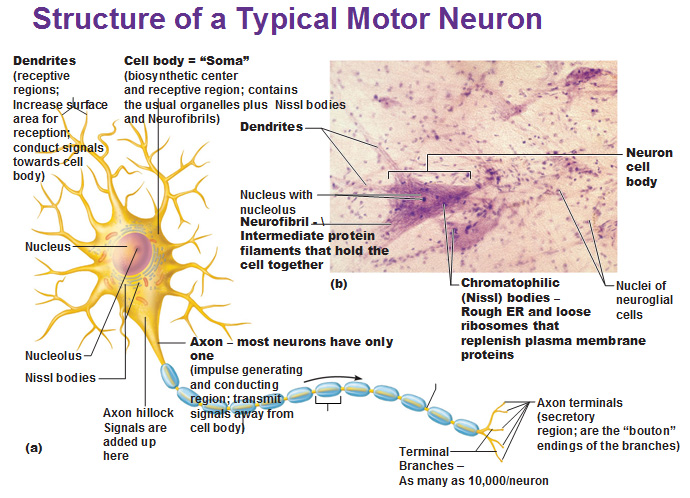
Clearly, new and improved treatment modalities are needed, but their development hinges on research platforms that faithfully reproduce the human pathology.ĭefects in the SCN1A gene, which encodes the α-subunit of the voltage-gated sodium channel Na v1.1, are seen in 70–80% of patients with DS, and approximately 50% of these defects truncate the Na v1.1 protein prematurely. Seizures are refractory to all currently available forms of treatment severe neuropsychiatric disabilities include cognitive deficits and autism-spectrum behaviors, and approximately 10–20% of the afflicted children do not survive. Our data indicate that patient-derived iPSCs may serve as a new and powerful research platform for genetic disorders, including the epilepsies.ĭravet syndrome (DS) is an infantile-onset epileptic encephalopathy that develops in a previously normal infant. Our results indicate a functional decline in Dravet neurons, especially in the GABAergic subtype, which supports previous findings in murine disease models, where loss-of-function in GABAergic inhibition appears to be a main driver in epileptogenesis. Current-clamp analyses revealed significant impairment in action potential generation when strong depolarizing currents were injected. Neurons derived from these iPSCs were primarily GABAergic (>50%), although glutamatergic neurons were observed as a minor population (<1%). We generated iPSCs from a Dravet syndrome patient with a c.4933C>T substitution in SCN1A, which is predicted to result in truncation in the fourth homologous domain of the protein (p.R1645*). We here report a cellular model for DS that utilizes patient-derived iPSCs. However, no such effort has been reported to date. Disease modeling using patient-derived induced pluripotent stem cells (iPSCs) can be a powerful tool to reproduce this syndrome’s human pathology. The achievement of electrosyringe-assisted electrospray MS is to unveil the molecular distribution in the whole living neuron, which offers the feasibility to deeply investigate molecular communication between the axon/dendrite and the body inside neurons.Dravet syndrome is a devastating infantile-onset epilepsy syndrome with cognitive deficits and autistic traits caused by genetic alterations in SCN1A gene encoding the α-subunit of the voltage-gated sodium channel Na v1.1. This uneven distribution is in accordance with the accumulation of neurotransmitters in the axon for information delivery. Higher levels of pyroglutamic acid and glutamic acid are revealed in the axon compared to in the body and dendrite. The ionization efficiency from the nano-capillary is enhanced to guarantee mass spectrometric analysis of multiple components from the axon and dendrite. The key is the insertion of a ∼130 nm capillary tip into one axon or dendrite to load the cytosol through electro-osmotic flow. Here, electrosyringe-assisted electrospray mass spectrometry (MS) is established for the first time to achieve intracellular sampling from one axon or dendrite in living neurons for mass spectrometric analysis. 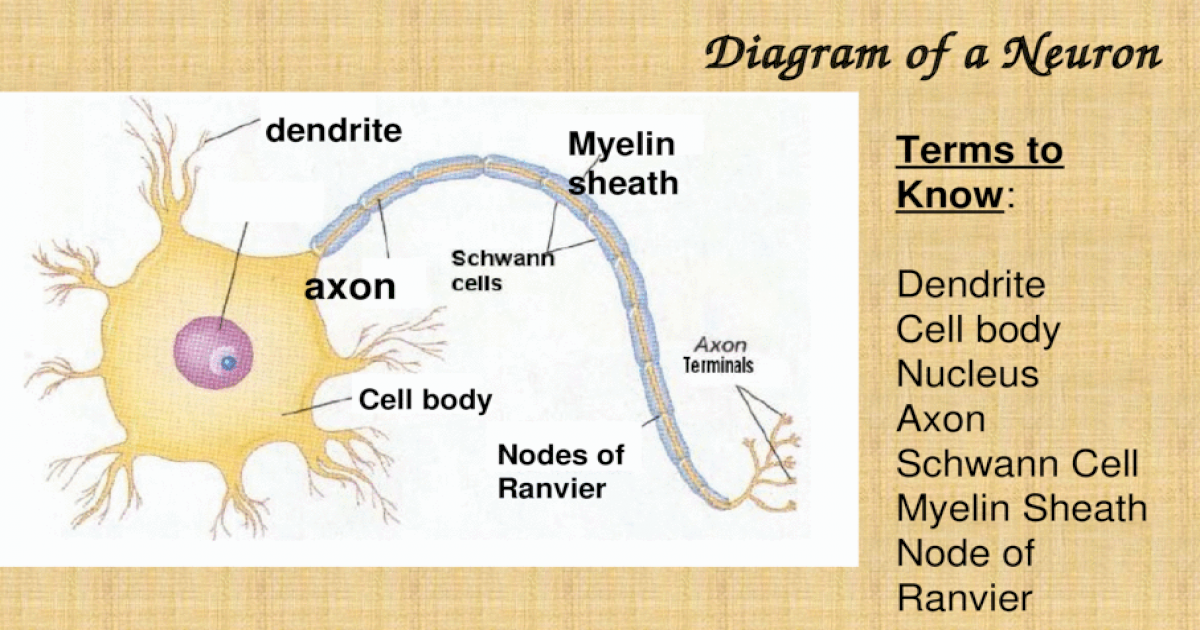
The molecular profiling of single axons and dendrites in living neurons could provide important information for the better understanding of neuron function. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
